Going on the offensive: The potential link between cell senescence and Alzheimer's
Cell senescence may trigger the start of cognitive decline. Can we stop it before it starts?
When you catch a cold or cut your finger, the immune system begins the healing process by clearing damaged cells.
As we age, that process weakens. Health problems linger as immune systems aren’t quite as robust as they once were, whether it’s fighting bacteria, viruses, or cancer cells. But there’s something beyond a cut healing more slowly that Alzheimer’s researchers are taking a closer look at.
One of these changes in the aging process is senescent cell accumulation. Senescent cells are cells throughout the body that have essentially lived too long and need to be replaced. Normally, these cells would degrade, and leftover pieces would help build new healthy cells. But senescent cells are stubborn. They block the normal degradation process, and the body can’t do anything to break them down.
“Somehow those cells send out certain signals to say ‘Hey, I’m kind of tired. Not only am I not going to do any more work, but I’m actually going to harm you,’” said Kevin Hascup, PhD, assistant professor at the Smith Alzheimer’s Center and the Department of Neurology at SIU School of Medicine.

Kevin Hascup, PhD
Maybe you’ve heard of senescent cells before, possibly in relation to cancer. It has also been linked to other conditions of aging, including diabetes and stroke. Similar thoughts of the relationship between cell senescence and disease have been applied to Alzheimer’s and dementia – if we’re able to address the issue of cell senescence, perhaps we can figure out how to develop treatments that prevent the accumulation of dangerous senescent cells or how those senescent cells trigger healthy cells in the body. Right now, it’s in the early stages of exploration.
“The buzzword — I don’t like the buzzword, but I see it a lot — they’re often referred to as zombie cells,” Hascup said. “They’re not living, but they’re not really dead. They’re in this in-between state.
“The problem is, they’re releasing a lot of unhealthy factors that go throughout the body and damage healthy cells as well.”
Reversing the damage?
While the research connecting cell senescence to Alzheimer's is in the early stages, the field of senescence is not new. The topic has been explored for more than 60 years, but the past decade has led to a better understanding of treatment.
This is where senolytic treatment comes in. Senolytics targets these senescent cells and kills them off, preventing any toxins they are releasing. So, the thought is by breaking up the accumulated senescent cells, we would be healthier and age-related issues would be less of a problem.
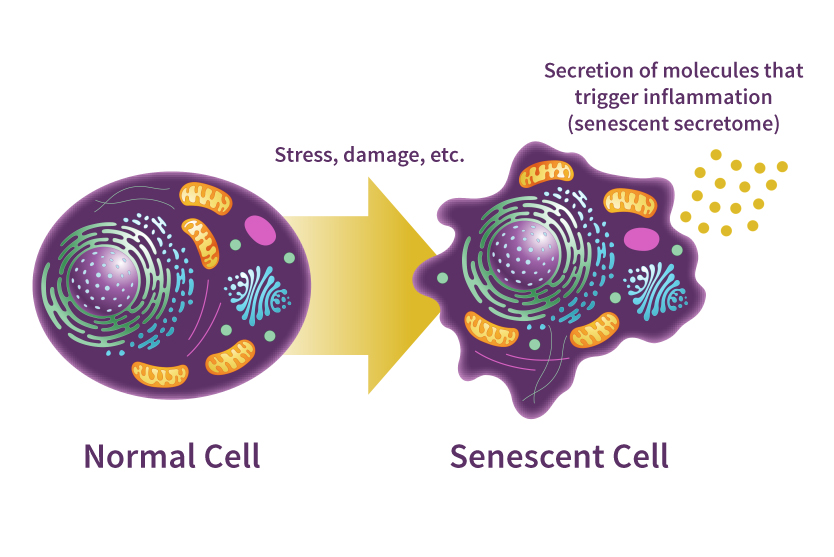
The difference between a normal and senescent cell,
courtesy of National Institute on Aging
The Hascup labs hypothesized – what if senolytics were administered before cell senescence accumulated? Could it improve healthy aging? Could you delay the onset of dementia?
They administered two different senolytic treatments to male and female mice. The first was Fisetin, and the second was a combination Quercetin and Dasatinib, the latter of which is a senolytic cancer treatment. All of these are plant-derived, coming from fruits and vegetables like strawberries, asparagus, onions and others. The idea is that it takes time to build up senescent cells, so one theory is it might be harmful to take treatment more often than needed. Treatments were given monthly.
The results were mixed. Male mice models responded favorably to the Fisetin treatment, but it had no effect on females. The females taking the Dasatinib and Quercetin combination experienced a detrimental effect, compared to male models who had minimal effects.
“Overall, we were pretty surprised with a majority of the findings,” Hascup said. “To see detrimental aspects in females for various aspects, including markers of inflammation, cognitive performance and metabolism — that was really the big one.”
This study focused on those in young adulthood, through a preventive medicine lens. Providing senolytic treatment in older models could offer up different responses yet again.
“Trying to get a better understanding of what we’re seeing in the Alzheimer’s disease models is almost reverse to what we saw in the natural aging models,” Hascup said. “Differences in genetics, different inflammatory states – all of that could play a big role.”
As with the vast majority of research, the findings don’t give one nice final answer. But it does point you in a better direction.
“One problem is that there’s no single test for senescence,” Hascup said. “Another problem is, that if you specifically state that you have senescent cells, it just indicates that you have inflammation. It could be from obesity, hypertension, MS, or a ton of other factors.”
Different gender responses
That difference in response from male and female mouse models has already created some buzz in the research world.
First, let’s back up. For a long time, the vast majority of research was only done with male models. It was 1) easier, and 2) cheaper considering you didn’t need to accommodate for sex differences. In 2016, the National Institute of Health (NIH), began instituting a new policy requiring grant applicants for preclinical studies to report the balance between male and female cells and animals. Since NIH is the world’s largest single funder of biomedical research, it made quite the ripple effect.

For Alzheimer’s and dementia, it seems logical to study the differences between men and women. Nearly two-thirds of Americans with Alzheimer’s are women, and while there are a number of theories why women are at a higher risk of developing Alzheimer’s than men, the difference is not completely explained.
The findings from the publication out of the Hascup lab raise another important reason – treatments may elicit different reactions. And just because one treatment may affect one group positively doesn’t mean that is the case for all.
“You separate the two sexes out, males may fare better with the drug, but females have no effect,” Hascup said. “We all talk about personalized medicine, but at the end of the day it’s a tricky thing to do.”
Senolytics, dementia and the future
Senolytics have risen in prominence thanks to its role in helping fight cancer cells, as well as its promise in treating other age-related diseases. Research like that usually leads to drug and supplement companies releasing products and making claims about the health benefits based on preliminary data that may be simply aspirational.
“There are some marketed as anti-aging compounds, but there’s very little literature showing how these compounds work in younger, healthier individuals,” Hascup said. “Caution is recommended in this emerging field.”
That’s why the Hascup lab is pushing its research to better understand the potential of senolytics, and its affect on all people. These senolytic treatments are barely a decade old, so understanding how their properties could help or hinder different diseases is still coming to light.
“In the field, there’s been a big effort put into understanding senescent cell accumulation. Understanding how that might impact aging, and how that might affect disease progression,” Hascup said. “These compounds have been routinely shown in older mice near the end of their life span and in various models that they provide very good benefits.
“If you’re a younger person taking this because you’ve seen an ad on TV, and think this will help me as I age, that might not actually be the case.”
There is hope, and there is certainly a better understanding of how different treatments may affect those with Alzheimer’s and dementia.
“If we can delay or slow the onset of biological aging, we have the opportunity to delay, prevent, or ameliorate chronic diseases.”



